|
Phys Rev B5:3988Īyers JE (2007) Heteroepitaxy of semiconductors. Jpn J Appl Phys 53:100201Īmmerlaan CAJ, Watkins GD (1972) Electron-paramagnetic-resonance detection of optically induced divacancy alignment in silicon. KeywordsĪkasaka T, Yamamoto H (2014) Nucleus and spiral growth mechanisms of nitride semiconductors in metalorganic vapor phase epitaxy. Planar defects comprise stacking faults, grain and twin boundaries, inversion-domain boundaries, and interfaces between different semiconductors or between a semiconductor and a metal. Dislocations are characterized by their Burgers vector and its angle to the dislocation line, and their mobility is provided by glide and climb processes. Most important are edge and screw dislocations, which affect crystal growth and accommodate strain in semiconductors. The mobility of defects is provided by various diffusion mechanisms and affected by their charge. Their creation is interrelated – among each other and also to the presence of extrinsic (impurity) defects – and governed by the conservation of particles and quasi-neutrality. Native (intrinsic) point defects and associates of these defects are formed at elevated temperature and may be frozen-in with decreasing temperature. Other defects promote nonradiative carrier recombination, carrier trapping, or excessive carrier scattering and are detrimental to device performance. These defects determine the desired electronic and optical properties of the semiconductor. Some defects are beneficial, such as donors, acceptors, or luminescence centers. They are classified into point, line, and planar defects. These are known as Bravias Lattices.Semiconducting properties of most interest are predominantly caused by crystal defects. There are fourteen types of lattices possible in a crystal. Q: How many kinds of space lattices are possible in a crystal?Īns: The correct option is “A”. End Centered: Has particles at the corners and one particle at the center of the opposite faces.Face Centered: This contains particles on every face of the lattice and other particles on the corners.Other particles (one or more) are at the corner of the lattice Body Centered: It has one particle at the center of the body.There are actually three types of non-primitive unit cells, namely: So there is more than one lattice point in a non-primitive unit cell. These additional constituent particles are either on the face of the unit cell or inside the unit cell. In this type of unit cell, there are particles not only at the corners of the lattice but in other positions as well. So essentially primitive unit cell has only one lattice point. There are no particles located at any other position in a primitive unit cell. The interfacial angles of the unit cell are as follows:Ī primitive unit cell only has atoms, molecules or ions at the corners of the lattice. C: edges defined by lattice vectors a and b.B: edges defined by lattice vectors a and c.A: edges defined by lattice vectors b and c.And these three edges form three respective angles. A unit cell is a geometric shape even by itself. The entire of the space lattice is built by the repeating arrangement of unit cells. It is used to visually simplify the crystalline patterns solids arrange themselves in. Every one of the fourteen lattices has such a unique geometryĪ unit cell is the most basic and least volume consuming repeating structure of any solid.By joining of these points we get the geometry (or shape) of the crystal.These lattice points of a crystal are joined together by straight lines.This particular particle may be an atom, a molecule or even ions.Each point on the lattice represents one particle of the crystal, This is a lattice point.The fourteen Bravais Lattices show some similar characteristics. The following diagram shows you the fourteen arrangements. These 14 arrangements are the Bravais Lattice. Now Auguste Bravais was French scientist who found out that there are a total of fourteen possible three-dimensional lattices. So a lattice is an array of points in a particular order which describes the arrangement of particles of a crystalline solid. When such an arrangement of atoms is represented in a three-dimensional structure, this is a crystal lattice. Let us take a look.Ĭrystals have a structure made up of a regular arrangement of their atoms (or particles). Another is a crystalline structure or crystals which have a specific organized structure of their particles. One is an amorphous solid which has no specific shape or structure. 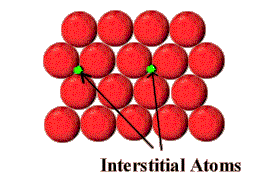
As we have studied in the previous topic, solids are basically of two shapes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
